 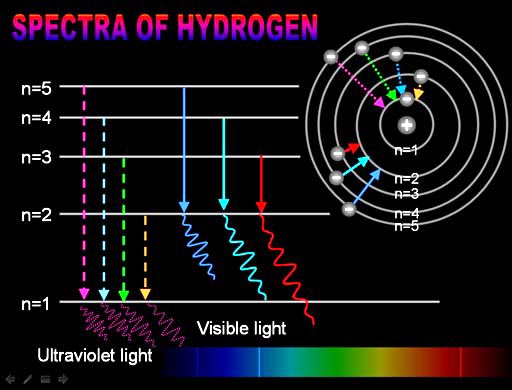
Plug in the value you have for #R# to get This implies that the Rydberg equation will take the form Now, you know that as the value of #n_2# increases, the value of #1/n_2^2# decreases. #n_2# represents the principal quantum number of the orbital that is higher in energy. 
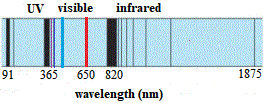
#n_1# represents the principal quantum number of the orbital that is lower in energy.#lamda_"e"# is the wavelength of the emitted photon (in a vacuum).Your tool of choice here will be the Rydberg equation for the hydrogen atom, which looks like this This transition is part of the Lyman series and takes place in the ultraviolet part of the electromagnetic spectrum. Therefore, each wavelength of the emission lines corresponds to an electron dropping from a certain energy level (greater than 1) to the first energy level.Start by calculating the wavelength of the emission line that corresponds to an electron that undergoes a #n=1 -> n = oo# transition in a hydrogen atom. Which is Rydberg's formula for the Lyman series. The version of the Rydberg formula that generated the Lyman series was: ġ λ = R H ( 1 − 1 n 2 ) ( R H ≈ 1.0968 × 10 7 m − 1 ≈ 13.6 eV h c ) Lyman-alpha radiation had previously been detected from other galaxies, but due to interference from the Sun, the radiation from the Milky Way was not detectable. On December 1, 2011, it was announced that Voyager 1 detected the first Lyman-alpha radiation originating from the Milky Way galaxy. Different versions of the Rydberg formula with different simple numbers were found to generate different series of lines. Rydberg managed to find a formula to match the known Balmer series emission lines, and also predicted those not yet discovered. Within five years Johannes Rydberg came up with an empirical formula that solved the problem, presented first in 1888 and final form in 1890. Nobody could predict the wavelengths of the hydrogen lines until 1885 when the Balmer formula gave an empirical formula for the visible hydrogen spectrum. Historically, explaining the nature of the hydrogen spectrum was a considerable problem in physics. Here is an illustration of the first series of hydrogen emission lines: The spectrum of radiation emitted by hydrogen is non-continuous or discrete. The rest of the lines of the spectrum (all in the ultraviolet) were discovered by Lyman from 1906-1914. The first line in the spectrum of the Lyman series was discovered in 1906 by physicist Theodore Lyman, who was studying the ultraviolet spectrum of electrically excited hydrogen gas. The greater the difference in the principal quantum numbers, the higher the energy of the electromagnetic emission. The series is named after its discoverer, Theodore Lyman. The transitions are named sequentially by Greek letters: from n = 2 to n = 1 is called Lyman-alpha, 3 to 1 is Lyman-beta, 4 to 1 is Lyman-gamma, and so on. In physics and chemistry, the Lyman series is a hydrogen spectral series of transitions and resulting ultraviolet emission lines of the hydrogen atom as an electron goes from n ≥ 2 to n = 1 (where n is the principal quantum number), the lowest energy level of the electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
